Abstract
Type 1 Gaucher disease (GD1) is a rare, autosomal recessive disorder. Deficient lysosomal glucocerebrosidase activity causes accumulation of glucosylceramide in macrophages that infiltrate bone marrow, spleen and liver. Common clinical manifestations include hematologic cytopenias, hepatosplenomegaly and bone lesions. Since 1991, GD1 has been treated primarily with pharmacological enzyme replacement therapy (ERT). In the pre-ERT era, sequelae of fibrotic liver disease were a common cause of early death. Ultrasonic and MR elastography (MRE) suggest that increased liver stiffness attributed to fibrosis correlates with GD1 disease severity scores. Here, we evaluate the impact of initial and post-ERT (median 20 y, percentiles5-95: 5.2-27.5 y) GD1 severity on probability for liver fibrosis (LF) in 30 patients (pts) using 4 well-established LF prediction scores. We also studied 9 never-treated pts and 7 pts treated for < 18 m and examined the relationship between LF, disease severity scores and D-dimer, ferritin, chitotriosidase and GBA1 genotype.
METHODS: 46 adult GD1 pts were evaluated in 2017-18. Pts with known liver disease or on anticoagulation were ineligible. Disease severity was scored with a validated assessment tool GD1-DS3: marked (DS3 6.0-19.0), moderate (DS3 3.0-5.99), mild (DS3 < 3.0). LF probability was assessed with AST/Platelet Ratio Index, Fibrosis-4 Index, Lok Score and NAFLD Fibrosis Score. Scores were classified positive, indeterminate or negative per published standards. Probabilities were stratified as: high (≥ 2 parameters positive), low (≥ 2 parameters negative), or indeterminate (any other score combination).
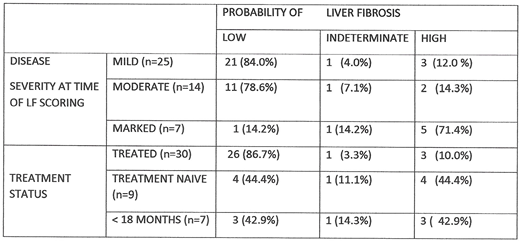
RESULTS: 26/30 pts (86.7%) treated for at least 3 y had low probability of LF at last evaluation. 4/9 (44.4%) ERT-naïve pts and 3/7 (42.9%) pts treated for ≤ 18 m had high probability for LF. 5/7 (71.4%) pts with marked DS3 severity had high LF probability compared to 2/14 (14.3%) with moderate DS3 severity and 3/25 (12.0%) with mild DS3 severity (Table). Of 30 pts treated for ≥ 3 y, 13 had marked DS3 severity scores before starting treatment. Only 1 pt had DS3≥6.00 at the time of last evaluation along with high probability for LF. She had a history of frequent and prolonged treatment interruptions as well as morbid obesity. Of 9 treated pts with moderate baseline severity scores, 4 remained so at last evaluation. One had high probability for LF but also had developed myelodysplasia and severe hemolytic anemia that likely confounded both GD1 severity and LF scores. The third treated pt with high probability for LF had a mild DS3 score but also insulin dependent diabetes mellitus and marked obesity. Chitotriosidase activity was >1000 nMol/mL/h in proportionately more pts with marked disease (5/7, 71.4%), compared to those with moderate (4/14, 28.6%) or mild disease (5/25, 20.0%) and among those pts who were never treated (8/9, 88.9%) or treated for <18 m (4/6, 66.7%), compared to those patients treated for ≥ 3 y. (11/31, 35.5%). All patients who were either untreated or treated for ≤ 18 m had ferritin levels >150 ng/mL, whereas ferritin was increased in less than half the patients treated ≥ 3 y. There was no correlation between these biomarkers and a high probability for LF. No patients had INR> 1.5 or abnormal AST or ALT. D-dimer levels did not correlate with disease severity or probability for LF. Of 10 patients with high probability for LF, 7 had platelet counts < 120x 103/µL. 8/17 (47.1%) with pre-treatment DS3> 6.0 were N370S homozygous compared to 23/28 (82.1%) with baseline moderate or mild severity scores. 7/10 patients with high probability for LF were N370S homozygous.
CONCLUSIONS: Per MRE studies reported by others, we find that LF probability based on non-invasive laboratory risk stratification is often increased parallel to GD1-DS3 score. However, in contrast to the pre-ERT era, most adult GD1 patients treated with ERT for a median 20 y, including those with initial marked disease severity, appear unlikely to develop LF. Exceptions may be attributed to confounding concurrent illnesses or to poor treatment compliance. Unlike DS3 score, we found no consistency between traditional GD1 biomarkers and probability for LF. We emphasize that N370S homozygosity can be associated with severe GD1 manifestations including LF and such pts should be treated accordingly. Our assessments of LF probability require verification with elastography especially in pts with thrombocytopenia.
Weinreb:Shire HGT: Honoraria, Membership on an entity's Board of Directors or advisory committees; Pfizer Corporation: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Genzyme, a Sanofi Company: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.